Date Log
Copyright (c) 2025 Jurnal Ilmiah Perikanan dan Kelautan

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.
1. Copyright of the article is transferred to the journal, by the knowledge of the author, whilst the moral right of the publication belongs to the author.
2. The legal formal aspect of journal publication accessibility refers to Creative Commons Atribusi-Non Commercial-Share alike (CC BY-NC-SA), (https://creativecommons.org/licenses/by-nc-sa/4.0/)
3. The articles published in the journal are open access and can be used for non-commercial purposes. Other than the aims mentioned above, the editorial board is not responsible for copyright violation
The manuscript authentic and copyright statement submission can be downloaded ON THIS FORM.
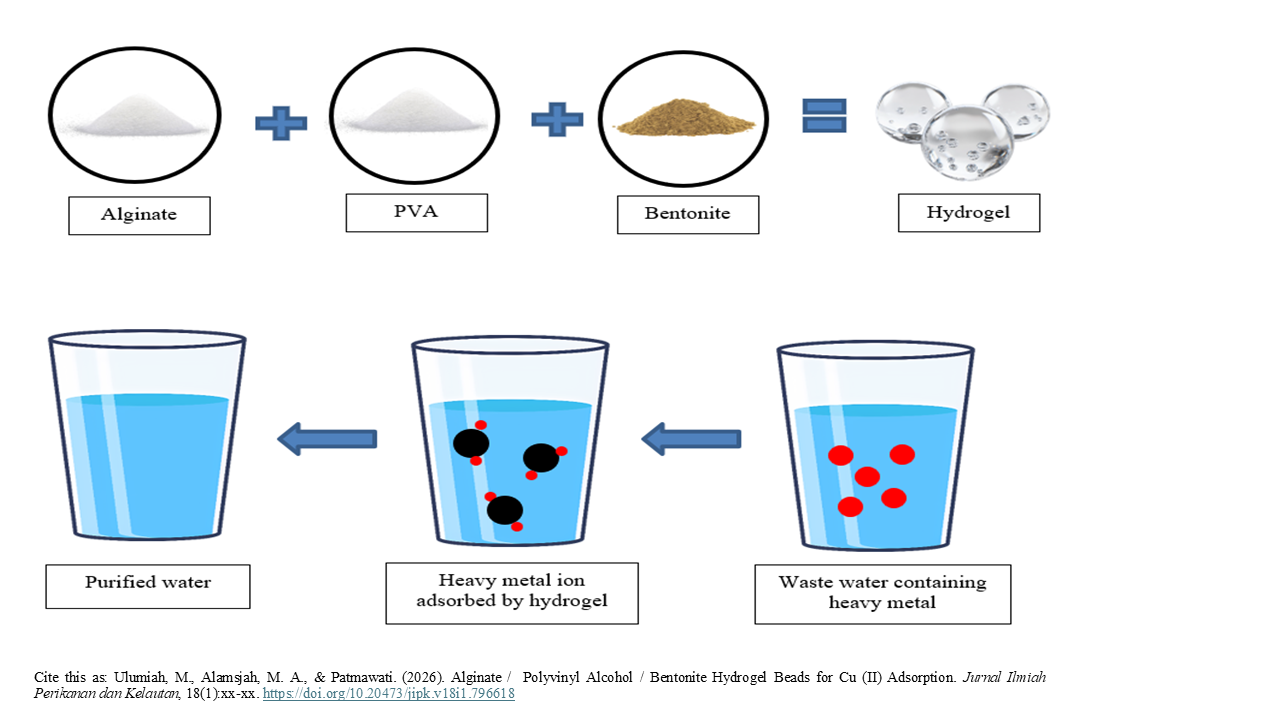
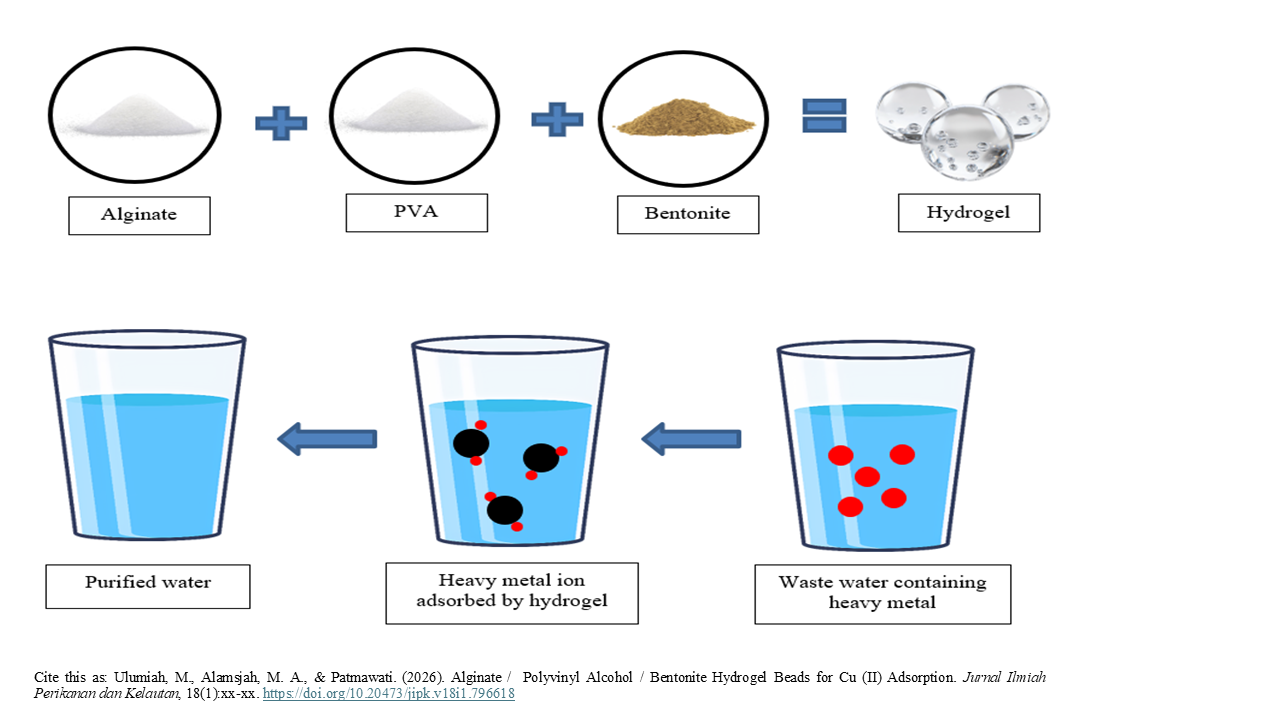
Alginate / Polyvinyl Alcohol / Bentonite Hydrogel Beads for Cu (II) Adsorption
Corresponding Author(s) : Mochammad Amin Alamsjah
Jurnal Ilmiah Perikanan dan Kelautan, 2025: IN PRESS ISSUE (JUST ACCEPTED MANUSCRIPT, 2025)
Abstract
Graphical Abstract
Highlight Research
- Hydrogel bead based on alginate, PVA and bentonite were prepared by ionic gelation.
- Hydrogel bead were tested on Cu standard solution and waste water from paper industry.
- Adding bentonite on hydrogel bead plays a key role for Cu removal.
- Hydrogel bead show selective removal on waste water containing Cu, Pb and Cr.
Abstract
Heavy metal pollution is a major problem for most water areas. One way to remove heavy metal ions is through the adsorption process by using combination of alginate / polyvinyl alcohol hydrogel. Adding bentonite can make good adsorption and effective. The purpose of this study was to analyze the effect of hydrogels for adsorption of heavy metals Cu (II) in liquid waste from paper industrial. This study used a Completely Randomized Design (CRD) experimental method with 5 treatments and 3 replications. Types of treatments include control treatments and the addition of bentonite with a ratio of 0.5 - 1.25%. The best treatment will be applied to liquid waste. The results showed that hydrogel beads from alginate / polyvinyl alcohol / bentonite have significant differences between treatments on the adsorption of heavy metal Cu (II). The best treatment was the addition of 1% bentonite (P4) with an adsorption capacity of 74.43%. The results of the application on liquid waste showed the following adsorption capacity: Cr (65.13%) > Cu (60.84%) > Pb (44.47%). Suggestions that can be given for further research are the application of hydrogel beads to other heavy metals that have the potential to increase adsorption capacity, such as Cr.
Keywords
Download Citation
Endnote/Zotero/Mendeley (RIS)BibTeX
- Aljar, M. A. A., Rashdan, S., & El-Fatttah, A. A. (2021). Environmentally friendly polyvinyl alcohol−alginate/ bentonite semi-interpenetrating polymer network nanocomposite hydrogel beads as an efficient adsorbent for the removal of methylene blue from aqueous solution. Polymers, 13(22):1-19.
- Azizah, M., & Mamay, M. (2021). Content of heavy metals lead (Pb), cadmium (Cd), and mercury (Hg) in the body of wader fish (Barbodes binotatus) and Cikaniki river water, Bogor Regency. Limnotek Perairan Darat Tropis di Indonesia, 28(2):83-93.
- Baigorria, E., Cano., L. A., Sanchez., L. M., Alvarez., V. A., & Ollier, R. P. (2020). Bentonite-composite polyvinyl alcohol/alginate hydrogel beads: Preparation, characterization and their use as arsenic removal devices. Environmental Nanotechnology, Monitoring & Management, 14(2):1-8.
- Bulin, C., Zheng, R., Guo, T., & Zhang, B. (2022). Incorporating hard-soft acid-base theory in multi-aspect analysis of the adsorption mechanism of aqueous heavy metals by graphene oxide. Journal of Physics and Chemistry of Solids, 170(11):1-10.
- Capan, M. E., & Cingoz, E. (2024). Hydrogel balls developed for use in the detection of heavy metals in wastewater. Journal of the Institute of Natural and Applied Sciences, 30(1):156-171.
- Chowdhury, N., Solaiman, Roy., C. K., Firoz, S. H., Foyez, T., & Imran, A. B. (2021). Role of ionic moieties in hydrogel networks to remove heavy metal ions from water. ACS Omega, 6(1):836-844.
- Darban, Z., Shahabuddin., S., Gaur, R., Ahmad, I., & Sridewi, N. (2022). Hydrogel-based adsorbent material for the effective removal of heavy metals from wastewater: A comprehensive review. Gels, 8(5):1-35.
- Ding, W., Liang, H., Zhang, H., Sun, Z., Geng, Z., & Xu, C. (2023). A cellulose/bentonite grafted polyacrylic acid hydrogel for highly-efficient removal of Cd (II). Journal of Water Process Engineering, 51(1):1-10.
- Du, Z., Liu, F., Xiao, C., Dan, Y., & Jiang, L. (2021). Fabrication of poly (vinyl alcohol) / sodium alginate hydrogel beads and its application in photo-fenton degradation of tetracycline. Journal of Materials Science, 56(1):913-926.
- Haryanto. (2021). The effect of chitosan on the characterization of PVA hydrogel films for wound dressing applications. Techno, 22(2):177-184.
- Harmesa, Lestari, & Budiyanto, F. (2020). Distribution of heavy metals in seawater and sediments in Cimanuk waters, West Java, Indonesia. Oseanologi dan Limnologi di Indonesia, 5(1):19-32.
- Heraldy, E., Purnamawati, N., Hidayat, Y., Noegrahaningtyas, K. D., & Nurcahyo, I. F. (2022). Preparation of biosorbent from kapok fruit peel (Ceiba pentandra) for adsorption of lead waste. Journal of Scientific and Applied Chemistry, 25(9):329-337.
- Hu, T., Liu, Q., Gao, T., Dong, K., Wei, G., & Yao, J. (2018). Facile preparation of tannic acid−poly(vinyl alcohol)/sodium alginate hydrogel beads for methylene blue removal from simulated solution. ACS Omega, 3(7):7523-7531.
- Isawi, H. (2020). Using zeolite/polyvinyl alcohol/sodium alginate nanocomposite beads for removal of some heavy metals from wastewater. Arabian Journal of Chemistry, 13(6):5691-5716.
- Jang, T., Yoon, S., Choi, J. H., Kim, N., & Park, J. A. (2025). Simultaneous removal of heavy metals and dyes on sodium alginate/polyvinyl alcohol/κ-carrageenan aerogel beads. Gels, 11(3):1-20.
- Kowalski, G., Witczak, M., & Kuterasinski, L. (2024). Structure effects on swelling properties of hydrogels based on sodium alginate and acrylic polymers. Molecules, 29(9):1-17.
- Kumar, S., & Jain, S. (2013). History, introduction, and kinetics of ion exchange materials. Journal of Chemistry, 2013(1):1-13.
- Lee, S. J., Lim, H. W., & Park, S. H. (2021). Adsorptive seawater desalination using MOF-incorporated Cu-alginate/PVA beads: Ion removal efficiency and durability. Chemosphere, 268(8):1-9.
- Li, X., Shu, M., Li, H., Gao, X., Long, S., Hu, T., & Wu, C. (2018). Strong, tough and mechanically self-recoverable poly (vinyl alcohol) / alginate dual-physical double-network hydrogels with large cross-link density contrast. RSC Advances, 8(30):16674-16689.
- Lin, Z., Yang, Y., Liang, Z., Zeng, L., & Zhang, A. (2021). Preparation of chitosan/calcium alginate/bentonite composite hydrogel and its heavy metal ions adsorption properties. Polymers, 13(11):1-19.
- Liu, Z., Zhao, J., Wang, A., Yuan, H., & Chi, Y. (2024). Adsorption behavior and mechanism of Cu (II) by sodium alginate/carboxymethyl cellulose/ magnesium hydroxide (SC-MH) hydrogel. International Journal of Biological Macromolecules, 277(48):1-11.
- Maulidina, R. F., Pujiani, D., & Haryanto. (2022). The effect of the addition of polyvinyl alcohol (PVA) concentrations on the characteristics of the carboxymethyl cellulose (CMC)-poly (acrylic acid) hydrogel superabsorbent as a planting medium. Chemica Jurnal Teknik Kimia, 9(2):60-66.
- Obsa, A., L., Shibeshi, N. T., Mulugeta, E., & Workeneh, G. A. (2025). Eco-friendly composite hydrogel based on cellulose and bentonite for removal of Lead (II) : Kinetics and isotherm studies. Carbohydrate Polymer Technologies and Applications, 9(1):1-15.
- Pathan, S., & Bose, S. (2018). Arsenic removal using “green” renewable feedstock-based hydrogels: Current and future perspectives. ACS Omega, 3(5):5910-5917.
- Prakash, J., Kumar, T. S., Venkataprasanna, K. S., Niranjan, R., Kaushik, M., Samal, D. B., & Venkatasubbu, G. D. (2019). PVA/alginate/hydroxyapatite films for controlled release of amoxicillin for the treatment of periodontal defects. Applied Surface Science, 495(32):1-9.
- Qasem, N., A. A., Mohammed, R. H., & Lawal, D. U. (2021). Removal of heavy metal ions from wastewater: A comprehensive and critical review. Clean Water, 4(36):1-15.
- Rao, W., Lv, G., Wang, D., & Liao, L. (2018). Enhanced degradation of Rh 6G by zero valent iron loaded on two typical clay minerals with different structures under microwave irradiation. Frontiers in Chemistry, 6(1):1-7.
- Rao, H., J. (2021). Characterization studies on adsorption of lead and cadmium using activated carbon prepared from waste tyres. Nature Environment and Pollution Technology, 20(2):561-568.
- Ratnasari, B. Y., Fadillah, N., Astuti, D. H., & Sani. (2021). Reduction of heavy metal ion levels in Karah River Water, Surabaya, using cation resin. Journal of Chemical and Process Engineering, 2(3):7-12.
- Saghandali, F., Salehi, M. B., & Taghikhani, V. (2023). Hydrogel nanocomposite network elasticity parameters as a function of swelling ratio: From micro to macro flooding. Journal of Industrial and Engineering Chemistry, 125(9):163-177.
- Sakr, M. A., Mohamed, M. G. A., Wu, R., Shin, S. R., Kim, D., Kim, K., & Siddiqua, S. (2020). Development of bentonite-gelatin nanocomposite hybrid hydrogels for tissue engineering. Applied Clay Science, 199(16):1-10.
- Santamaria, D. E. G., Justel, A., Fernandez, R., Ruiz, A. I., Stavropoulou, A., Blanco, J. D. R., & Cuevas, J. (2021). SEM-EDX study of bentonite alteration under the influence of cement alkaline solutions. Applied Clay Science, 212(13):1-16.
- Shen, Q., Wu, T., Zhang, G. B., Ma, H. E., Wang, W., Pan, G. X., & Zhang, Y. F. (2023). Synthesis of magnetic bentonite–gelatin hydrogel beads and their applications in Cu2+ capturing. Environmental Science and Pollution Research, 30(60):125702-125717.
- Stachurski, Z. H. (2011). On structure and properties of amorphous materials. Materials, 4(9):1564-1598.
- Surdu, V. A., & Gyorgy, R. (2023). X-ray diffraction data analysis by machine learning methods - a review. Applied Science, 13(17):1-22.
- Syeda, H. I., Sultan, I., Razavi, K. S., & Yap, P. S. (2022). Biosorption of heavy metals from aqueous solution by various chemically modified agricultural wastes: A review. Journal of Water Process Engineering, 46(2):1-42.
- Tammina, S. K., Priyadarshi, R., Khan, A., Manzoor, A., Rahman, R. S. H. A., & Banat, F. (2025). Recent developments in alginate-based nanocomposite coatings and films for biodegradable food packaging applications. International Journal of Biological Macromolecules, 295(13):1-20.
- Tuljanah, N., Ahmad, A., Sondari, D., Septevani, A. A., Amanda, P., Hanifah, A. S., Karim, A., & Karim, H. (2024). Properties of hydrogel for adsorbent textile dye waste based cellulose-carboxymethyl sago starch. IOP Conference Series: Earth and Environmental Science, 1388(1):1-14.
- Wang, T., Zhang, F., Zhao, R., Wang, C., Hu, K., Sun, Y., Politis, C., Shavandi, A., & Nie, L. (2020). Polyvinyl alcohol/sodium alginate hydrogels incorporated with silver nanoclusters via green tea extract for antibacterial applications. Designed Monomers and Polymers, 23(1):118-133.
- Wang, Z., Li, T. T., Peng, H. K., Reng, H. T., Lou, C. W., & Lin, J. H. (2021). Low-cost hydrogel adsorbent enhanced by trihydroxy melamine and β-cyclodextrin for the removal of Pb (II) and Ni (II) in water. Journal of Hazardous Materials, 411(12):1-10.
- Wang, P., Shen, X., Qiu, S., Zhang, L., Ma, Y., & Liang, J. (2024). Clay-based materials for heavy metals adsorption: Mechanisms, advancements, and future prospects in environmental remediation. Crystals, 14(12):1-18.
- Windri, R., E. (2011). Analysis of Cu (II) Content with Residual and Sulfate Ions using a Visible Spectrophotometer in Raw Water and Refilled Drinking Water in Pekanbaru City. Sultan Syarif Kasim State Islamic University, Riau, 92 pg.
- Yi, X., Sun, F., Han, Z., Han, F., He, J., Ou, M., Gu, J., & Xu, X. (2018). Graphene oxide encapsulated polyvinyl alcohol/sodium alginate hydrogel microspheres for Cu (II) and U (VI) removal. Ecotoxicology and Environmental Safety, 158(12):309-318.
- Zhang, B., Cui, Y., Yin, G., & Li, X. (2012). Adsorption of copper (II) and lead (II) ions onto cottonseed protein-PAA hydrogel composite. Polymer-Plastics Technology and Engineering, 51(6):612-619.
- Zhang, W., Deng, Q., He, Q., Song, J., Zhang, S., Wang, H., Zhou, J., & Zhang, H. (2018). A facile synthesis of core-shell/bead-like poly (vinyl alcohol)/alginate@ PAM with good adsorption capacity, high adaptability and stability towards Cu (Ⅱ) removal. Chemical Engineering Journal, 351(21):462-472.
- Zhixing, X., Lijuan, Z., Long, W.,& Dan, C. (2019). Adsorptive removal of Cu (II) from aqueous solutions using a novel macroporous bead adsorbent based on poly (vinyl alcohol)/sodium alginate/KMNO4 modified biochar. Journal of the Taiwan Institute of Chemical Engineers, 102(9):110-117.
- Zhu, H., Chen, S., & Luo, Y. (2023). Adsorption mechanisms of hydrogels for heavy metal and organic dyes removal: A short review. Journal of Agriculture and Food Research, 12(2):1-5.
- Zhumagaliyeva, S. N., Iminovа, R. S., Kairalapova, G. Z., Beysebekov, M. M., Beysebekov, M. K., & Abilov, Z. A. (2017). Composite polymer-clay hydrogels based on bentonite clay and acrylates: Synthesis, characterization and swelling capacity. Eurasian Chemico-Technological Journal, 19(3):279-288.
References
Aljar, M. A. A., Rashdan, S., & El-Fatttah, A. A. (2021). Environmentally friendly polyvinyl alcohol−alginate/ bentonite semi-interpenetrating polymer network nanocomposite hydrogel beads as an efficient adsorbent for the removal of methylene blue from aqueous solution. Polymers, 13(22):1-19.
Azizah, M., & Mamay, M. (2021). Content of heavy metals lead (Pb), cadmium (Cd), and mercury (Hg) in the body of wader fish (Barbodes binotatus) and Cikaniki river water, Bogor Regency. Limnotek Perairan Darat Tropis di Indonesia, 28(2):83-93.
Baigorria, E., Cano., L. A., Sanchez., L. M., Alvarez., V. A., & Ollier, R. P. (2020). Bentonite-composite polyvinyl alcohol/alginate hydrogel beads: Preparation, characterization and their use as arsenic removal devices. Environmental Nanotechnology, Monitoring & Management, 14(2):1-8.
Bulin, C., Zheng, R., Guo, T., & Zhang, B. (2022). Incorporating hard-soft acid-base theory in multi-aspect analysis of the adsorption mechanism of aqueous heavy metals by graphene oxide. Journal of Physics and Chemistry of Solids, 170(11):1-10.
Capan, M. E., & Cingoz, E. (2024). Hydrogel balls developed for use in the detection of heavy metals in wastewater. Journal of the Institute of Natural and Applied Sciences, 30(1):156-171.
Chowdhury, N., Solaiman, Roy., C. K., Firoz, S. H., Foyez, T., & Imran, A. B. (2021). Role of ionic moieties in hydrogel networks to remove heavy metal ions from water. ACS Omega, 6(1):836-844.
Darban, Z., Shahabuddin., S., Gaur, R., Ahmad, I., & Sridewi, N. (2022). Hydrogel-based adsorbent material for the effective removal of heavy metals from wastewater: A comprehensive review. Gels, 8(5):1-35.
Ding, W., Liang, H., Zhang, H., Sun, Z., Geng, Z., & Xu, C. (2023). A cellulose/bentonite grafted polyacrylic acid hydrogel for highly-efficient removal of Cd (II). Journal of Water Process Engineering, 51(1):1-10.
Du, Z., Liu, F., Xiao, C., Dan, Y., & Jiang, L. (2021). Fabrication of poly (vinyl alcohol) / sodium alginate hydrogel beads and its application in photo-fenton degradation of tetracycline. Journal of Materials Science, 56(1):913-926.
Haryanto. (2021). The effect of chitosan on the characterization of PVA hydrogel films for wound dressing applications. Techno, 22(2):177-184.
Harmesa, Lestari, & Budiyanto, F. (2020). Distribution of heavy metals in seawater and sediments in Cimanuk waters, West Java, Indonesia. Oseanologi dan Limnologi di Indonesia, 5(1):19-32.
Heraldy, E., Purnamawati, N., Hidayat, Y., Noegrahaningtyas, K. D., & Nurcahyo, I. F. (2022). Preparation of biosorbent from kapok fruit peel (Ceiba pentandra) for adsorption of lead waste. Journal of Scientific and Applied Chemistry, 25(9):329-337.
Hu, T., Liu, Q., Gao, T., Dong, K., Wei, G., & Yao, J. (2018). Facile preparation of tannic acid−poly(vinyl alcohol)/sodium alginate hydrogel beads for methylene blue removal from simulated solution. ACS Omega, 3(7):7523-7531.
Isawi, H. (2020). Using zeolite/polyvinyl alcohol/sodium alginate nanocomposite beads for removal of some heavy metals from wastewater. Arabian Journal of Chemistry, 13(6):5691-5716.
Jang, T., Yoon, S., Choi, J. H., Kim, N., & Park, J. A. (2025). Simultaneous removal of heavy metals and dyes on sodium alginate/polyvinyl alcohol/κ-carrageenan aerogel beads. Gels, 11(3):1-20.
Kowalski, G., Witczak, M., & Kuterasinski, L. (2024). Structure effects on swelling properties of hydrogels based on sodium alginate and acrylic polymers. Molecules, 29(9):1-17.
Kumar, S., & Jain, S. (2013). History, introduction, and kinetics of ion exchange materials. Journal of Chemistry, 2013(1):1-13.
Lee, S. J., Lim, H. W., & Park, S. H. (2021). Adsorptive seawater desalination using MOF-incorporated Cu-alginate/PVA beads: Ion removal efficiency and durability. Chemosphere, 268(8):1-9.
Li, X., Shu, M., Li, H., Gao, X., Long, S., Hu, T., & Wu, C. (2018). Strong, tough and mechanically self-recoverable poly (vinyl alcohol) / alginate dual-physical double-network hydrogels with large cross-link density contrast. RSC Advances, 8(30):16674-16689.
Lin, Z., Yang, Y., Liang, Z., Zeng, L., & Zhang, A. (2021). Preparation of chitosan/calcium alginate/bentonite composite hydrogel and its heavy metal ions adsorption properties. Polymers, 13(11):1-19.
Liu, Z., Zhao, J., Wang, A., Yuan, H., & Chi, Y. (2024). Adsorption behavior and mechanism of Cu (II) by sodium alginate/carboxymethyl cellulose/ magnesium hydroxide (SC-MH) hydrogel. International Journal of Biological Macromolecules, 277(48):1-11.
Maulidina, R. F., Pujiani, D., & Haryanto. (2022). The effect of the addition of polyvinyl alcohol (PVA) concentrations on the characteristics of the carboxymethyl cellulose (CMC)-poly (acrylic acid) hydrogel superabsorbent as a planting medium. Chemica Jurnal Teknik Kimia, 9(2):60-66.
Obsa, A., L., Shibeshi, N. T., Mulugeta, E., & Workeneh, G. A. (2025). Eco-friendly composite hydrogel based on cellulose and bentonite for removal of Lead (II) : Kinetics and isotherm studies. Carbohydrate Polymer Technologies and Applications, 9(1):1-15.
Pathan, S., & Bose, S. (2018). Arsenic removal using “green” renewable feedstock-based hydrogels: Current and future perspectives. ACS Omega, 3(5):5910-5917.
Prakash, J., Kumar, T. S., Venkataprasanna, K. S., Niranjan, R., Kaushik, M., Samal, D. B., & Venkatasubbu, G. D. (2019). PVA/alginate/hydroxyapatite films for controlled release of amoxicillin for the treatment of periodontal defects. Applied Surface Science, 495(32):1-9.
Qasem, N., A. A., Mohammed, R. H., & Lawal, D. U. (2021). Removal of heavy metal ions from wastewater: A comprehensive and critical review. Clean Water, 4(36):1-15.
Rao, W., Lv, G., Wang, D., & Liao, L. (2018). Enhanced degradation of Rh 6G by zero valent iron loaded on two typical clay minerals with different structures under microwave irradiation. Frontiers in Chemistry, 6(1):1-7.
Rao, H., J. (2021). Characterization studies on adsorption of lead and cadmium using activated carbon prepared from waste tyres. Nature Environment and Pollution Technology, 20(2):561-568.
Ratnasari, B. Y., Fadillah, N., Astuti, D. H., & Sani. (2021). Reduction of heavy metal ion levels in Karah River Water, Surabaya, using cation resin. Journal of Chemical and Process Engineering, 2(3):7-12.
Saghandali, F., Salehi, M. B., & Taghikhani, V. (2023). Hydrogel nanocomposite network elasticity parameters as a function of swelling ratio: From micro to macro flooding. Journal of Industrial and Engineering Chemistry, 125(9):163-177.
Sakr, M. A., Mohamed, M. G. A., Wu, R., Shin, S. R., Kim, D., Kim, K., & Siddiqua, S. (2020). Development of bentonite-gelatin nanocomposite hybrid hydrogels for tissue engineering. Applied Clay Science, 199(16):1-10.
Santamaria, D. E. G., Justel, A., Fernandez, R., Ruiz, A. I., Stavropoulou, A., Blanco, J. D. R., & Cuevas, J. (2021). SEM-EDX study of bentonite alteration under the influence of cement alkaline solutions. Applied Clay Science, 212(13):1-16.
Shen, Q., Wu, T., Zhang, G. B., Ma, H. E., Wang, W., Pan, G. X., & Zhang, Y. F. (2023). Synthesis of magnetic bentonite–gelatin hydrogel beads and their applications in Cu2+ capturing. Environmental Science and Pollution Research, 30(60):125702-125717.
Stachurski, Z. H. (2011). On structure and properties of amorphous materials. Materials, 4(9):1564-1598.
Surdu, V. A., & Gyorgy, R. (2023). X-ray diffraction data analysis by machine learning methods - a review. Applied Science, 13(17):1-22.
Syeda, H. I., Sultan, I., Razavi, K. S., & Yap, P. S. (2022). Biosorption of heavy metals from aqueous solution by various chemically modified agricultural wastes: A review. Journal of Water Process Engineering, 46(2):1-42.
Tammina, S. K., Priyadarshi, R., Khan, A., Manzoor, A., Rahman, R. S. H. A., & Banat, F. (2025). Recent developments in alginate-based nanocomposite coatings and films for biodegradable food packaging applications. International Journal of Biological Macromolecules, 295(13):1-20.
Tuljanah, N., Ahmad, A., Sondari, D., Septevani, A. A., Amanda, P., Hanifah, A. S., Karim, A., & Karim, H. (2024). Properties of hydrogel for adsorbent textile dye waste based cellulose-carboxymethyl sago starch. IOP Conference Series: Earth and Environmental Science, 1388(1):1-14.
Wang, T., Zhang, F., Zhao, R., Wang, C., Hu, K., Sun, Y., Politis, C., Shavandi, A., & Nie, L. (2020). Polyvinyl alcohol/sodium alginate hydrogels incorporated with silver nanoclusters via green tea extract for antibacterial applications. Designed Monomers and Polymers, 23(1):118-133.
Wang, Z., Li, T. T., Peng, H. K., Reng, H. T., Lou, C. W., & Lin, J. H. (2021). Low-cost hydrogel adsorbent enhanced by trihydroxy melamine and β-cyclodextrin for the removal of Pb (II) and Ni (II) in water. Journal of Hazardous Materials, 411(12):1-10.
Wang, P., Shen, X., Qiu, S., Zhang, L., Ma, Y., & Liang, J. (2024). Clay-based materials for heavy metals adsorption: Mechanisms, advancements, and future prospects in environmental remediation. Crystals, 14(12):1-18.
Windri, R., E. (2011). Analysis of Cu (II) Content with Residual and Sulfate Ions using a Visible Spectrophotometer in Raw Water and Refilled Drinking Water in Pekanbaru City. Sultan Syarif Kasim State Islamic University, Riau, 92 pg.
Yi, X., Sun, F., Han, Z., Han, F., He, J., Ou, M., Gu, J., & Xu, X. (2018). Graphene oxide encapsulated polyvinyl alcohol/sodium alginate hydrogel microspheres for Cu (II) and U (VI) removal. Ecotoxicology and Environmental Safety, 158(12):309-318.
Zhang, B., Cui, Y., Yin, G., & Li, X. (2012). Adsorption of copper (II) and lead (II) ions onto cottonseed protein-PAA hydrogel composite. Polymer-Plastics Technology and Engineering, 51(6):612-619.
Zhang, W., Deng, Q., He, Q., Song, J., Zhang, S., Wang, H., Zhou, J., & Zhang, H. (2018). A facile synthesis of core-shell/bead-like poly (vinyl alcohol)/alginate@ PAM with good adsorption capacity, high adaptability and stability towards Cu (Ⅱ) removal. Chemical Engineering Journal, 351(21):462-472.
Zhixing, X., Lijuan, Z., Long, W.,& Dan, C. (2019). Adsorptive removal of Cu (II) from aqueous solutions using a novel macroporous bead adsorbent based on poly (vinyl alcohol)/sodium alginate/KMNO4 modified biochar. Journal of the Taiwan Institute of Chemical Engineers, 102(9):110-117.
Zhu, H., Chen, S., & Luo, Y. (2023). Adsorption mechanisms of hydrogels for heavy metal and organic dyes removal: A short review. Journal of Agriculture and Food Research, 12(2):1-5.
Zhumagaliyeva, S. N., Iminovа, R. S., Kairalapova, G. Z., Beysebekov, M. M., Beysebekov, M. K., & Abilov, Z. A. (2017). Composite polymer-clay hydrogels based on bentonite clay and acrylates: Synthesis, characterization and swelling capacity. Eurasian Chemico-Technological Journal, 19(3):279-288.