Pathogenicity of Clostridium perfringens Philippine Isolate in Necrotic Enteritis Across Broiler Growth Stages
Downloads
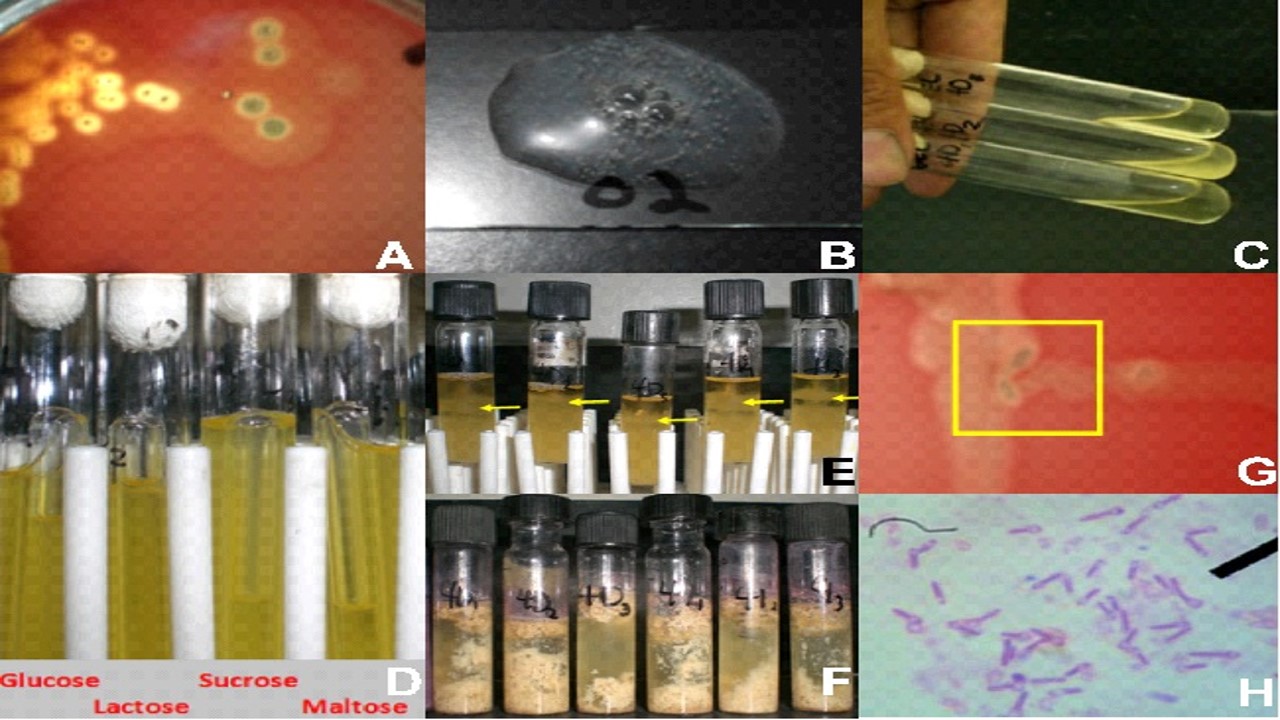
Clostridium perfringens was isolated from Philippines broiler chickens of a local farm exhibiting clinical signs of necrotic enteritis. This local isolate induced necrotic enteritis (NE) experimentally in susceptible broiler chickens to demonstrate the disease and the lesions it would produce. Experimental chickens were subjected to stress such as vaccination and pathogenic Escherichia coli and Salmonella enteritidis. Seven treatment groups involving various combinations of the above agents were used to demonstrate lesions of NE. Gross intestinal lesion scoring was performed at necropsy on the 3rd, 7th, 11th, 14th, and 18th-day post-infection, depending on the treatment group. Results showed that the local C. perfringens isolate was able to cause lesions of enteritis but did not demonstrate the classic towel-like lesion of NE, as described by the literature. The treatment combination of C. perfringens and S. enteritidis produced the highest intestinal lesions scores. However, C. perfringens alone can experimentally induce enteritis with a lesser severity. This is the first report of experimental induction of NE in broiler chickens using a local C. perfringens isolate in the Philippines.
Adhikari, P., Kiess, A., Adhikari, R., & Jha, R. (2020). An approach to alternative strategies to control avian coccidiosis and necrotic enteritis. Journal of Applied Poultry Research, 29(2), 515–534.
Alnassan, A. A., Kotsch, M., Shehata, A. A., Kruger, M., Daugschies, A., & Bangoura, B. (2014). Necrotic enteritis in chickens: developmentof a straightforward disease model system. The Veterinary Record, 1–7.
Chang, S., Kwak, W., Lee, J., Kim, S., Song, D., An, J., Park, S., Jeon, K., Kim, H., & Cho, J. (2024). Effect of stimbiotic on growth performance, nutrient digestibility, oocyst shedding, blood profiles, and intestinal microbiota in necrotic enteritis-challenged broiler. Animal Biotechnology, 35(1).
Craven, S. E., Stern, N. J., Bailey, J. S., & Cox, N. A. (2001). Incidence of Clostridium perfringens in broiler chickens and their environment during production and processing. Avian Diseases, 45(4), 887–896.
Fathima, S., Hakeem, W. G. Al, Shanmugasundaram, R., & Selvaraj, R. K. (2022). Necrotic Enteritis in Broiler Chickens: A Review on the Pathogen, Pathogenesis, and Prevention. Microorganisms, 10(10), 1–29.
Feng, X., Li, T., Zhu, H., Liu, L., Bi, S., Chen, X., & Zhang, H. (2022). Effects of challenge with Clostridium perfringens, Eimeria and both on ileal microbiota of yellow feather broilers. Frontiers in Microbiology, 13(December), 1–13.
Gao, C. H., Cao, H., Cai, P., & Sorensen, S. J. (2021). The initial inoculation ratio regulates bacterial coculture interactions and metabolic capacity. The ISME Journal, 15, 29–40.
Goossens, E., Valgaeren, B. R., Pardon, B., Haesebrouck, F., Ducatelle, R., Deprez, P. R., & Immerseel, F. V. (2017). Rethinking the role of alpha toxin in Clostridium perfringens-associated enteric diseases: a review on bovine necro-haemorrhagic enteritis. Veterinary Research, 48(9).
Habib Wani, A., Hussain, I., Maqbool, R., Dar, P. S., India, S., Ganaie, M. Y., Wani, S. A., Wani, A. H., Hussain, I., Kashoo, Z. A., & Qureshi, S. (2017). Isolation, identification and molecular characterization of Clostridium perfringens from poultry in Isolation, identification and molecular characterization of Clostridium perfringens from poultry in Kashmir valley, India. Journal of Entomology and Zoology Studies, 5(5), 409–414.
Hafez, H. M., & Attia, Y. A. (2020). Challenges to the Poultry Industry: Current Perspectives and Strategic Future After the COVID-19 Outbreak. Frontiers in Veterinary Science, 7(August).
Hanif, H., Anjum, A., Ali, N., Jamal, A., Imran, M., Ahmad, B., & Ali, M. I. (2015). Isolation and antibiogram of Clostridium tetani from clinically diagnosed tetanus patients. American Journal of Tropical Medicine and Hygiene, 93(4), 752–756.
Hofacre, C. L., Smith, J. A., & Mathis, G. F. (2018). An optimist’s view on limiting necrotic enteritis and maintaining broiler gut health and performance in today’s marketing, food safety, and regulatory climate. Poultry Science, 97(6), 1929–1933.
Jesudhasan, P. R., Bhatia, S. S., Sivakumar, K. K., Praveen, C., Genovese, K. J., He, H. L., Droleskey, R., McReynolds, J. L., Byrd, J. A., Swaggerty, C. L., Kogut, M. H., Nisbet, D. J., & Pillai, S. D. (2021). Controlling the Colonization of Clostridium perfringens in Broiler Chickens by an Electron-Beam-Killed Vaccine. Animals (Basel), 11(3).
Jiang, Y., Kulkarni, R. R., Parreira, V. R., Poppe, C., Roland, K. L., & Prescott, J. F. (2010). Assessment of 2 Salmonella enterica serovar Typhimurium-based vaccines against necrotic enteritis in reducing colonization of chickens by Salmonella serovars of different serogroups. Canadian Journal of Veterinary Research, 74(4), 264–270.
Kartikasari, A. M., Hamid, I. S., Purnama, M. T. E., Damayanti, R., Fikri, F., & Praja, R. N. (2019). Isolation And Identification Of Escherichia Coli As Bacterial Contamination In Broiler Chicken Meat In Poultry Slaughterhouse Lamongan District. Jurnal Medik Veteriner, 2(1), 66–71.
Keyburn, A. L., Bannam, T. L., Moore, R. J., & Rood, J. I. (2010). Net B, a Pore-Forming Toxin from Necrotic Enteritis Strains of Clostridium Perferingens. Toxins, 2(7), 1913–1927.
Keyburn, A. L., Sheedy, S. A., Ford, M. E., Williamson, M. M., Awad, M. M., Rood, J. I., & Moore, R. J. (2006). Alpha-toxin of Clostridium perfringens is not an essential virulence factor in necrotic enteritis in chickens. Infection and Immunity, 74(11), 6496–6500.
Khabiri, A., Toroghi, R., Mohammadabadi, M., & Tabatabaeizadeh, S. (2022). Cloning and nucleotide sequencing of the complete matrix protein of Newcastle disease virus subgenotype VII. 1.1 prevalence in broiler flocks of northeastern Iran. Journal of Genetics, 17(2), 113–125.
Khabiri, A., Toroghi, R., Mohammadabadi, M., & Tabatabaeizadeh, S. E. (2023). Introduction of a Newcastle disease virus challenge strain (sub-genotype VII. 1.1) isolated in Iran. Veterinary Research Forum, 14(4), e221.
Lee, K. W. & Lillehoj, H. S. (2021). Role of Clostridium perfringens Necrotic Enteritis B-like Toxin in Disease Pathogenesis. Vacciness (Basel), 10(1).
M’Sadeq, S. A., Wu, S., Swick, R. A., & Choct, M. (2015). Towards the control of necrotic enteritis in broiler chickens with in-feed antibiotics phasing-out worldwide. Animal Nutrition, 1(1), 1–11.
Maluping, R. P., Villanueva, R. R., & Vizmanos, M. F. C. (2003). Isolation of Clostridium perfringens from the duodenum of broilers with enteritis from selected farms in Southern Luzon, Philippines. Philippine Journal of Veterinary Medicine, 40(1), 46–48.
Moazeni, S., Mohammadabadi, M. R., Sadeghi, M. Shahrbabak, H. M., Koshkoieh, A. E., & Bordbar, F. (2016a). Association between UCP Gene Polymorphisms and Growth, Brreeding Value of Growth and Reproductive Traits in Mazandaran Indigenous Chicken. Open Journal of Animal Science, 6, 1–8.
Moazeni, S. M., Mohammadabadi, M. R., Sadeghi, M., Shahrbabak, H. M., & Esmailizadeh, A. (2016b). Association of the melanocortin-3(MC3R) receptor gene with growth and reproductive traits in Mazandaran indigenous chicken. Journal of Livestock Science and Technologies, 4(2), 51–56.
Mohammadifar, A, & Mohammadabadi, M. R. (2018) Melanocortin-3 receptor (MC3R) gene association with growth and egg production traits in Fars indigenous chicken. Malaysian Applied Biology, 47(3), 85–90.
Mohammadifar, A., & Mohammadabadi, M. R. (2017). Effect of uncoupling protein polymorphisms on growth, breeding value of growth and reproductive traits in the fars indigenous chicken. Iranian Journal of Applied Animal Science, 7(4), 679–685.
Mohiuddin, M., Yuan, W., Song, Z., Liao, S., Qi, N., Li, J., Lv, M., Wu, C., Lin, X., Hu, J., Cai, H., & Sun, M. (2021). Experimental induction of necrotic enteritis with or without predisposing factors using netB positive Clostridium perfringens strains. Gut Pathogens, 13(1), 1–7.
Natalie Berkhout. (2020). Philippines: Chicken overtakes pork as preferred protein. Poultry World.
Navarro, M. A., McClane, B. A., & Uzal, F. A. (2018). Mechanisms of action and cell death associated with Clostridium perfringens toxins. Toxins, 10(5), 1–21.
Okaiyeto, S. A., Sutar, P. P., Chen, C., Mi, J. B., Wang, J. Mujumdar, A. S., Zhang, J. S., Xu, M. Q., Fang, X. M., Zhang, C., & Xiao H. W. (2024). Antibiotic resistant bacteria in food systems: Current status, resistance mechanisms, and mitigation strategies. Agriculture Communications, 2(1).
Olkowski, A. A., Wojnarowicz, C., Chirino-Trejo, M., & Drew, M. D. (2006). Responses of broiler chickens orally challenged with Clostridium perfringens isolated from field cases of necrotic enteritis. Research in Veterinary Science, 81(1), 99–108.
Peek, S.., McGuirk, S. M., Sweeney, R. W., & Cummings, K. J. (2018). Infectious Diseases of the Gastrointestinal Tract. In Diseases of Body System, pp: 249–356.
Raman, M., Banu, S. S., Gomathinayagam, S., & Raj, G. D. (2011). Lesion scoring technique for assessing the virulence and pathogenicity of Indian field isolates of avian Eimeria species. Veterinarski Arhiv, 81(2), 259–271.
Shahdadnejad, N., Mohammadabadi, M. R., & Shamsadini, M. (2016). Typing of Clostridium Perfringens Isolated from Broiler Chickens Using Multiplex PCR. Genetics in the Third Millennium, 14(4), 4368–4374.
Copyright (c) 2025 Ma. Rosario S. Racho, Listya Purnamasari, Joseph P. Olarve, Hope G. Rovira, Joseph Flores dela Cruz

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.
Authors who publish in this journal agree to the following terms:
1. The journal allows the author to hold the copyright of the article without restrictions;
2. The journal allows the author(s) to retain publishing rights without restrictions;
3. The legal formal aspect of journal publication accessibility refers to Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License (CC BY-NC-SA).






11.jpg)